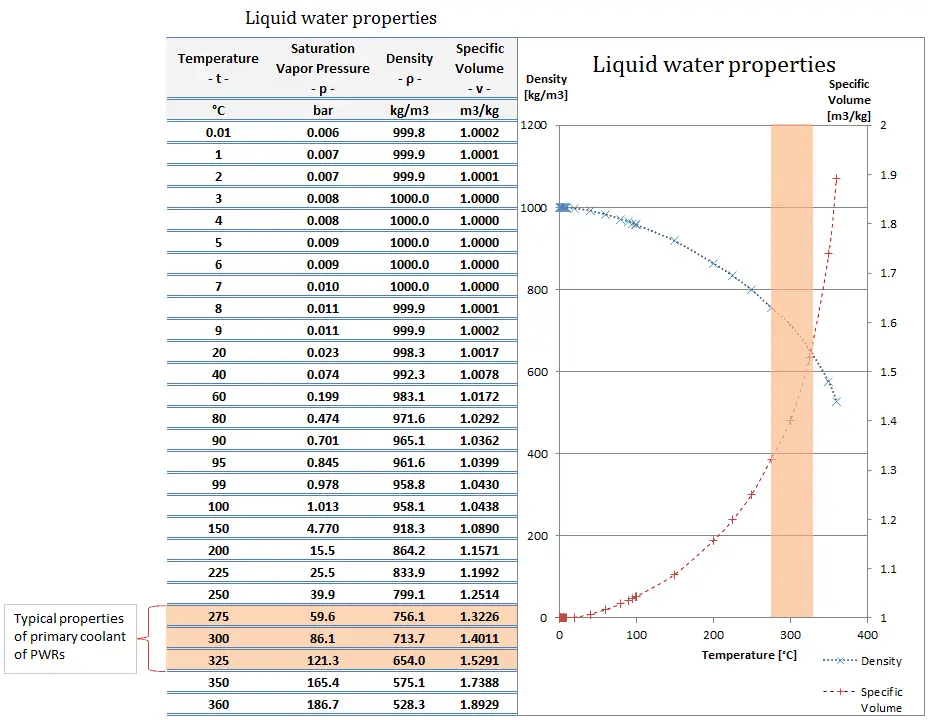 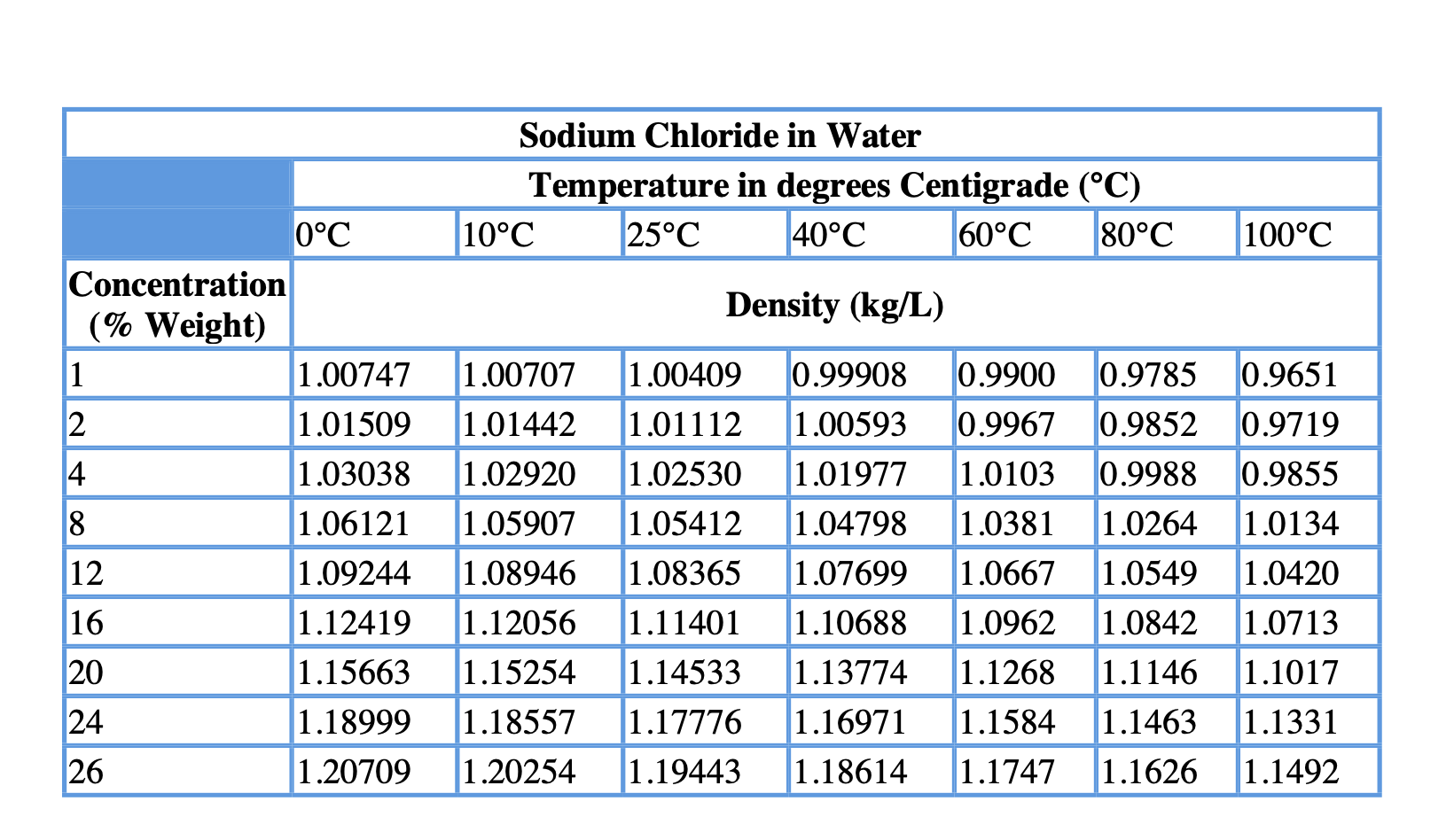
The results are in tabular form and interpolated into graphic form. 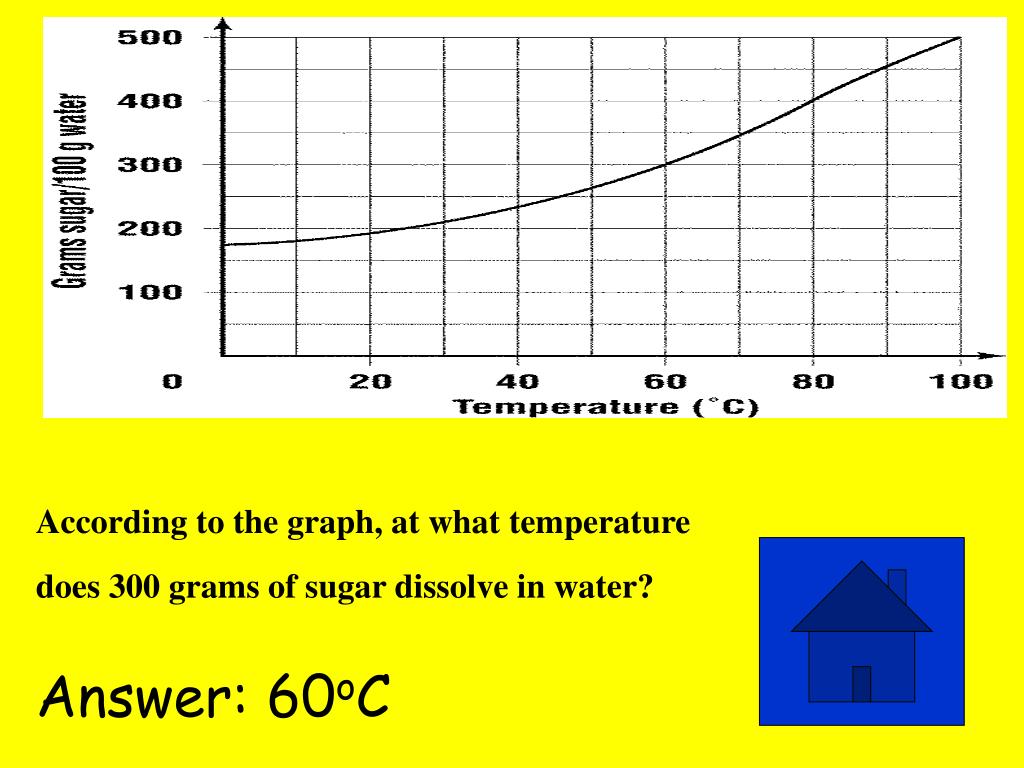
It is produced naturally in plants and is the main constituent of white sugar.It has the molecular formula C 12 H 22 O 11. This can be converted to calories or to joules easily. Sucrose, a disaccharide, is a sugar composed of glucose and fructose subunits. From the molarity, the number of calories/ gal can be calculated. Question 2: A solution of water and salt contains 25 grams of salt in 250 mL of water. Answer 1: The sugar cube has a density of 1.4 grams/cm 3. This helps calculate colligative properties such as freezing point or boiling point of any solution. Step 2: Plug your variables into the density formula. The molarity (m) measured the moles of solute per kilogram of solvent. To measure the mass of the fluid, you’ll need a balance. This makes comparisons between solutions easy. To calculate the density of sugar water, for example, you will need a graduated cylinder. The molarity (M) is a normalized method of measuring the moles of solute per volume of final solution. Density (or the similar specific gravity) is measured easily in the field. We have then measured the density, molarity, molality, calories/gal and freezing point of various ratios. Density of some sugars, alcohols and other organic substances in water is plotted as function of wt, mol/kg water and mol/l solution. We have made up a series of sugar (sucrose, Imperial Pure Cane granulated) solutions using the percentage volumes of water and dry sugar. Add 2 level tablespoons (26 g or 3 sugar cubes) of granulated sugar to the first cup, 4 to the second cup, 6 to the third cup and 8 tablespoons to the fourth cup. In either case it is not easy to compare one recipe to another as far as energy content or total amount of sugar. For one bound water per sucrose, the molar. Most people using sugar water for hummingbird feeders use a recipe ratio involving volumes of water (solvent) to volumes of dry granular sugar (solute) or they measure the final volume of the solution instead of added solvent. While no information is available for FOS, data for sucrose 41 allow an estimation of500 Å 3 per fully hydrated molecule, with five waters per sucrose. & Biochem., Angela State University, San Angelo, TX. Physical and chemical properties of various sugar water ratios for hummingbird feeders.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
